 March 04, 2026
March 04, 2026Looking back at 2024, the medical device industry can truly be described as undergoing rapid, transformative change.
Volume-based procurement (VBP) continues to advance intensively; numerous so-called "forbidden zones" previously considered off-limits to centralized procurement have been breached one after another, while the implementation of winning bids has come under comprehensive regulatory oversight. A steady stream of new healthcare policies has been introduced—measures such as consumable traceability codes, direct settlement between medical insurance authorities and enterprises, and the mutual recognition of diagnostic test results—which have precisely targeted and dismantled long-standing systemic ills within the medical device sector. Concurrently, official industry regulations are being updated in lockstep with these developments.
The anti-corruption campaign within the healthcare sector remains in full swing; the tendering and bidding processes—which are inextricably linked to medical devices—have been repeatedly singled out by the Central Commission for Discipline Inspection (CCDI). Furthermore, issues such as grassroots-level corruption and medical insurance fraud are receiving heightened scrutiny. The large-scale equipment renewal initiative, launched in early 2024, is now poised to enter a phase of accelerated volume growth; procurement demand that had been temporarily deferred is expected to surge rapidly into the market this year.
As 2025 arrives, a new industry cycle is officially unfolding.
01
Centralized Procurement:
Intensive Implementation + Strict Enforcement
The highly anticipated fifth round of national centralized procurement for medical consumables officially opened for bidding last December. The unit price for cochlear implant consumables plummeted from an average of over 200,000 yuan to approximately 50,000 yuan, while the winning bids for peripheral vascular stents generally hovered between 3,000 and 4,000 yuan—representing a significant price reduction across the board compared to previous levels.
According to the National Healthcare Security Administration (NHSA), the implementation of the winning bids for cochlear implants is expected to commence around March 3, 2025. The implementation of winning bids for peripheral vascular interventional consumables is scheduled to take effect in May 2025.
Among the seven major categories of medical consumables prioritized for national centralized procurement under the guidance of the National Healthcare Security Administration (NHSA), the tenders for Fujian’s ligation clips, Jiangxi’s biochemical reagents, Guangdong’s ultrasonic scalpel tips, Zhejiang’s breast biopsy needles, and Anhui’s tumor markers and thyroid function tests have already been opened. Furthermore, the tenders for Henan’s specialized coronary balloons and Hebei’s vascular interventional devices are also nearing their opening dates. The relevant winning bid results for all these categories are expected to be successively implemented throughout 2025.
According to the NHSA’s *Notice on Strengthening Regional Coordination to Enhance the Quality and Scope of Centralized Pharmaceutical Procurement in 2024*, the centralized procurement of high-value medical consumables should focus on product categories characterized by inflated prices, significant market representativeness, or strong public concern. Furthermore, provincial-level consortium procurements that meet the necessary criteria should be elevated to the status of national-level consortium procurements.
It is foreseeable that, building upon existing provincial-level consortium procurements, several new national-level centralized procurement initiatives for medical consumables may emerge in 2025. (A summary of provincial-level consortium procurements conducted in 2024 can be found at the end of this article.)
Regarding the implementation of centralized procurement results, the NHSA and the National Health Commission (NHC) jointly announced ten specific measures last December. These measures cover various stages—including the hospital admission, utilization, and monitoring of centrally procured drugs and consumables—and are expected to play a pivotal role in the implementation phase of various centralized procurement initiatives throughout 2025. Key measures include:
Starting from the third month following the implementation of each batch of centralized procurement results, relevant authorities must organize a round of inspections to identify issues regarding the hospital admission status of winning drugs and consumables within their respective regions. They are to urge medical institutions that have not yet completed the admission and procurement process for these winning products to do so as soon as possible. Additionally, authorities are required to issue periodic public reports identifying provinces and specific product categories that exhibit significant delays in procurement progress, an excessively high proportion of non-winning drugs and consumables in their procurement volume, or other related issues.
02
Policies and Regulations:
Enhanced Safeguards + Stricter Regulation
· **Nationwide Rollout of Consumable Traceability Codes; Upgraded Price Monitoring Mechanisms**
The National Healthcare Security Administration (NHSA) issued a document today emphasizing that, effective January 1, 2025, it will fully implement a rigorous regulatory regime anchored by the use of traceability codes.
For pharmaceutical and medical consumable enterprises, these traceability codes serve as a tool to monitor the production, transportation, and sales of their products in real time, enabling the timely detection and resolution of issues. Furthermore, enterprises can use the codes to track terminal market prices, thereby avoiding the negative repercussions that may arise for manufacturers when products are sold below their officially listed prices.
Should regulatory authorities discover quality defects in any pharmaceuticals or consumables, the traceability codes allow them to quickly pinpoint the specific affected batches and their distribution status, thereby facilitating timely product recalls and corrective actions.
Through the seamless integration of traceability codes with healthcare insurance settlement services, the healthcare security authorities can effectively prevent fraudulent activities—such as the illicit resale, substitution, or duplicate billing of insured pharmaceuticals and consumables—thereby safeguarding the integrity of the healthcare insurance fund.
· **Accelerated Implementation of Direct Settlement and Advance Payments; Improved Payment Collection Environment for the Pharmaceutical and Medical Device Industries**
Prior to the Lunar New Year, the NHSA convened a national symposium on healthcare security administration in Boao, Hainan. During the meeting, it was emphasized that efforts must be actively undertaken to promote the system of advance fund payments, as well as to advance the implementation of immediate, direct, and synchronized settlement mechanisms.
To date, numerous regions across the country have initiated direct healthcare insurance settlements specifically for pharmaceuticals and consumables procured through centralized volume purchasing. Statistical data from various localities—along with information disclosed by the NHSA—indicate that this initiative has resulted in a significant reduction in the payment collection cycle for pharmaceutical and consumable suppliers. The scope of direct healthcare insurance settlements is expected to expand further in 2025.
A joint notice issued by the NHSA and the National Health Commission (NHC)—titled *Notice on Improving the Working Mechanisms for Centralized Volume Purchasing and Implementation of Pharmaceuticals and Medical Devices*—stipulates that priority shall be given to advancing unified settlement through provincial-level platforms for products procured via centralized purchasing, while also exploring the possibility of extending this mechanism to other categories of pharmaceuticals and medical devices. Recently, Wenchang District in Hainan Province launched a pilot program to incorporate non-centralized procurement consumables used in local public hospitals into the scope of direct settlement.
A prepayment system for medical insurance funds was implemented last November. This system helps designated medical institutions alleviate the financial pressure associated with fronting medical expenses—specifically, the working capital required for expenditures such as the procurement of pharmaceuticals and medical consumables.
According to the official schedule, designated medical institutions may voluntarily apply to the medical insurance authorities in their respective unified planning areas for these advance payments during the first ten days of January each year.
· **Timeline Established for Mutual Recognition of Diagnostic Results; 2025 Targets Announced**
According to the *Guiding Opinions on Further Promoting the Mutual Recognition of Diagnostic and Laboratory Results Among Medical Institutions*—issued jointly by the National Health Commission and six other government departments—by the end of 2025, all "tightly-knit" medical alliances (including urban medical groups and county-level medical communities) are expected to achieve mutual recognition for all diagnostic and laboratory items among the institutions within their respective networks. Furthermore, within each prefecture-level city, the number of mutually recognized items among local medical institutions is expected to exceed 200.
As the scope of mutual recognition continues to expand, the volume of unnecessary diagnostic and laboratory procedures will steadily decrease, thereby further curtailing the prevalence of over-treatment.
· **Key Regulations Nearing Implementation: Medical Devices, Pharmaceutical Representatives, and More**
In August 2024, the Department of Comprehensive Affairs at the National Medical Products Administration (NMPA) released a draft of the *Law of the People's Republic of China on the Administration of Medical Devices (Draft for Public Comment)* to solicit public feedback. Comprising 11 chapters and 190 articles, the draft covers every stage of the medical device lifecycle—including research and development, manufacturing, distribution, and utilization—as well as various related aspects such as standards, classification, vigilance, and product recalls. The public consultation phase for this draft has now concluded, and the legislation is expected to officially come into force in 2025.
Furthermore, the newly revised *Regulations on the Supervision and Administration of Medical Devices* will come into effect on January 20, 2025. The key revisions are as follows:

On November 28, 2024, the National Medical Products Administration (NMPA) released the *Administrative Measures for Pharmaceutical Representatives (Draft for Comments)*. These measures shall take effect from the date of issuance, at which time the *Administrative Measures for the Filing of Pharmaceutical Representatives (Trial Implementation)* shall be simultaneously repealed. To date, the draft has solicited feedback from six government agencies—including the Ministry of Public Security, the National Health Commission, the State Administration for Market Regulation, the National Healthcare Security Administration, the National Administration of Traditional Chinese Medicine, and the National Administration of Disease Control and Prevention—signaling that its official implementation is fast approaching.
The *Draft for Comments* reiterates the definition of a pharmaceutical representative and, for the first time, explicitly establishes the qualification requirements for the position. Article 10 of the draft stipulates that a pharmaceutical representative must hold a bachelor's degree or higher in medicine, pharmacy, or a related field (or possess an intermediate-level or higher professional technical title). Furthermore, they must possess theoretical knowledge of clinical pharmacology and practical experience in the pharmaceutical field—or have work experience in areas such as drug R&D, manufacturing, testing, or quality management. They are required to master knowledge regarding the pharmacology and toxicology, functions and indications (or approved uses), combination therapies, adverse reactions, contraindications, and precautions associated with the drugs they promote; additionally, they must have successfully completed training and passed the assessment administered by the respective Drug Marketing Authorization Holder.
03
Medical Anti-Corruption:
Targeted Upgrades + Deepening Reach at the Grassroots Level
Today (January 2), the official website of the Central Commission for Discipline Inspection (CCDI) published an article titled *Promoting High-Quality Development of Disciplinary Inspection and Supervision Work on the New Journey: Resolutely Rectifying Malpractices and Corruption Issues Affecting the Public*. The article highlights that corruption within the pharmaceutical sector exacerbates the financial burden on the public, damages the relationship between medical professionals and patients, and degrades the overall professional ethos of the industry.
In 2024, the high-pressure stance against corruption in the healthcare sector continued unabated. According to CCTV News, the Central Commission for Discipline Inspection and the National Supervisory Commission (CCDI-NSC) mobilized disciplinary inspection and supervisory agencies at all levels to coordinate with relevant departments in launching a heavy-handed crackdown. They rigorously investigated corrupt practices—such as "profiting from one's medical position" and the fraudulent misappropriation of medical insurance funds—and resolutely curbed industry malpractices, such as accepting kickbacks and "red envelopes" (bribes). Nationwide, a total of 52,000 individuals were placed under investigation, 40,000 received disciplinary sanctions, and 2,634 were referred to procuratorial organs for criminal prosecution.
According to incomplete statistics compiled by Saibailan, at least 136 hospital presidents and Party secretaries across various levels of hospitals were publicly reported this year for suspected disciplinary and legal violations; they are currently undergoing disciplinary reviews and supervisory investigations by the CCDI-NSC or have already received disciplinary sanctions.
Among the cases reported recently, there has been a marked increase in the exposure of "micro-corruption"—petty corruption occurring at the grassroots level. Previously, the *Work Plan of the Central Anti-Corruption Coordination Group (2023–2027)*—issued by the General Office of the CPC Central Committee—explicitly stated that the state would push for anti-corruption efforts to extend down to the grassroots level.
From December 30 to 31, 2024, the 2025 National Health and Wellness Work Conference was held in Beijing. The conference noted that 2025 marks the final year of the "14th Five-Year Plan" period. It emphasized the need to strengthen integrity building and vigorously deepen the rectification of improper conduct and corrupt practices that directly affect the public.
04
Industry Development:
Market Recovery + Innovation Going Global
With the vast potential of overseas markets and intensifying domestic competitive pressures, "going global" has emerged as the optimal solution for medical device enterprises seeking to expand their business growth. It is also an essential path for leading brands to achieve internationalization and global reach.
On December 31, 2024, CICC (34.890, 0.00, 0.00%) released a research report stating that, although listed medical device companies have experienced some earnings volatility in recent quarters—influenced by factors such as ongoing industry-wide rectifications, slow progress in equipment procurement and bidding, and centralized procurement initiatives for IVD and high-value consumables—it has also been observed that the marginal impact of these factors is gradually diminishing or showing signs of recovery. Consequently, specific sub-sectors within the industry are expected to benefit from product upgrades and overseas exports in 2025.
According to statistics from SDIC Securities, the scale of medical equipment tenders and bids in the first half of 2024 amounted to approximately 55 billion yuan, representing a year-on-year decline of about 35%. Within this total, the bidding volume for medical imaging equipment fell by roughly 45% year-on-year, while the volume for the life information sector (patient monitoring and support systems) declined by 50% to 60%.
Currently, procurement demand that had previously been delayed due to equipment replacement cycles is gradually being released. According to statistics from Zhongcheng Digital, as of November 17 of last year, the total budgeted amount for intended equipment replacement purchases in the medical sector reached 17.7 billion yuan. Notably, in November alone, both the budgeted amount and the number of projects for medical equipment replacement saw a significant increase, signaling that relevant policies have now entered the implementation phase.
Everbright Securities (17.180, -0.10, -0.58%) projects that the current wave of equipment replacement initiatives will drive approximately 60 billion yuan in medical equipment procurement, potentially propelling medical device manufacturers into a period of high earnings growth. Furthermore, domestic medical device suppliers are continuously achieving innovative breakthroughs, and government policies are actively supporting the localization of high-end medical equipment. Given that the current domestic substitution rates remain relatively low in fields such as electrophysiology, endoscopy, CT imaging, and high-end ultrasound, policy support is expected to accelerate the market penetration of domestically produced products, thereby ushering the industry into a new phase of growth.
As intense market competition heats up, the importance of product innovation has become increasingly prominent. Currently, regions such as Beijing, Shanghai, Anhui, Shandong, and Hubei have successively introduced measures to create "fast lanes" for the innovative development of medical devices, covering specific details ranging from market access and clinical application to payment channels and fiscal-financial support.
Amidst an increasingly competitive and saturated domestic market environment, "going global" to tap into overseas opportunities has become a key strategic priority for numerous medical device enterprises. According to data compiled by Yizhuang Shusheng, China's medical device trade maintained an export surplus in the first half of 2024, with exports of both medical consumables and IVD (In Vitro Diagnostics) products growing by over 6%. It is projected that the total export value of medical devices for the full year will exceed that of the previous year, marking a return to export trade growth after a three-year hiatus.
Based on the financial reports released by various companies in 2024, domestic brands have already achieved significant results in areas such as breakthroughs in high-end equipment and the widespread rollout of low-value consumables; this progress is expected to accelerate further in 2025.
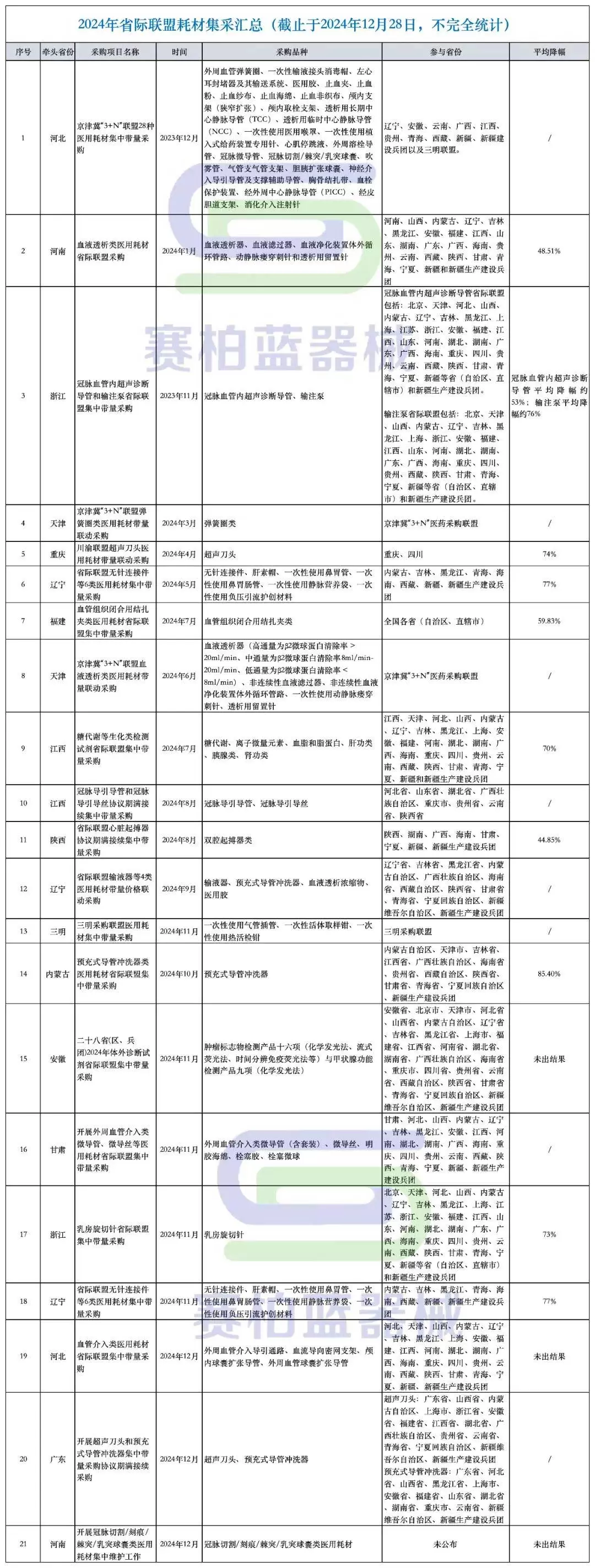
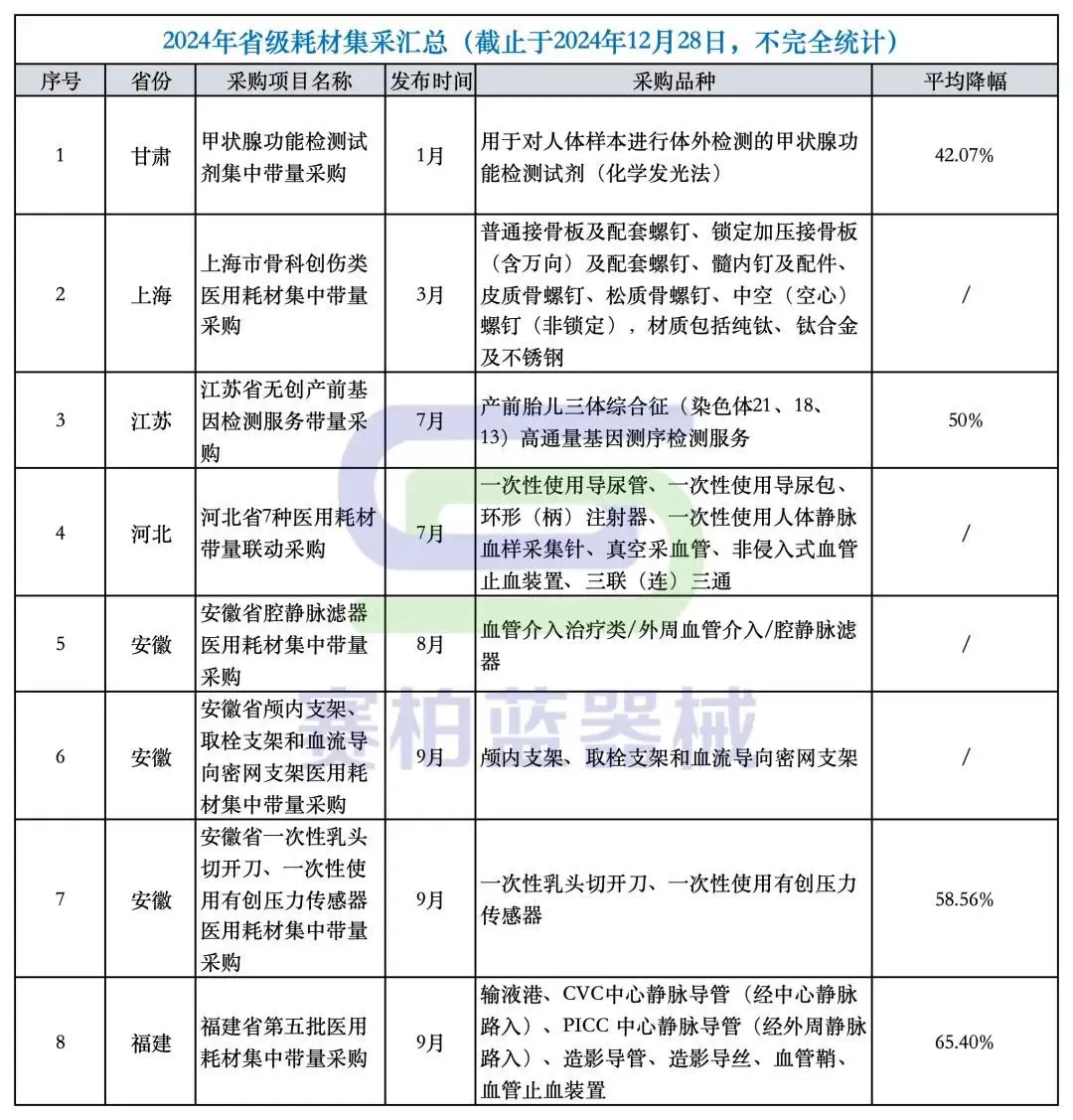
Source: Tencent.com

 March 04, 2026
March 04, 2026
 March 04, 2026
March 04, 2026
 March 04, 2026
March 04, 2026
 March 04, 2026
March 04, 2026Encountering problems or challenges? Please contact us!