 March 02, 2026
March 02, 2026On November 24, the Beijing Municipal Bureau of Economy and Information Technology, together with five other government departments, jointly issued the *Measures of Beijing Municipality for Promoting the High-Quality Development of the Medical Device Industry* (hereinafter referred to as the "Measures"). These Measures introduce 15 specific policies across five major dimensions—R&D and registration, commercialization of results, ecosystem clustering, digital empowerment, and open cooperation—covering nearly the entire value chain of medical device industry development. The initiative aims to enable Beijing's medical device industry to achieve both "innovative breakthroughs" and "deep-rooted growth," thereby establishing a high-end industrial hub with global influence. This policy comes into effect on November 24, 2025, and remains valid until December 31, 2028. Notably, the *Measures* serve as an "accelerator" for the "AI + Healthcare" sector; they encourage medical institutions to incorporate novel AI-assisted diagnostic and treatment technologies into their clinical practice by referencing existing pricing categories for AI-based medical services.

For medical device enterprises, the most vexing challenges often revolve around the "difficulty of R&D, slow time-to-market, and struggles with commercialization." The newly released *Measures* precisely target these pain points, establishing a "full-chain" support system for enterprises that spans from the initial source to the final end-user. The *Measures* encourage enterprises, research institutions, and medical institutions to form interdisciplinary innovation consortia that bridge the fields of medicine and engineering. Through the implementation of the "Capital Medical Technology Innovation Commercialization Promotion Plan" and public roadshows for the "Innovating for the Future" project, the initiative aims to drive the practical application and commercialization of forward-looking innovative achievements. Regarding approval speeds—specifically addressing the pain point of "slow time-to-market" for urgently needed medical devices—the policy explicitly mandates immediate acceptance and inspection upon submission. Consequently, the inspection timeframe for passive medical devices and diagnostic reagents has been reduced to an average of 60 working days, while the timeframe for active medical devices has been compressed to an average of 90 working days.
To facilitate the commercialization and widespread adoption of innovative medical devices—thereby bringing high-quality products from the laboratory into the hospital—the *Measures* propose the timely inclusion of eligible innovative medical devices into the *Zhongguancun Innovative Medical Device Product Catalog* and the *Beijing Catalog of First-of-its-Kind Major Technical Equipment*. Furthermore, the *Measures* call for intensified promotional efforts directed at healthcare institutions, the organization of regular product showcase events, and the refinement of procedures and mechanisms for the direct recommendation and admission of cataloged products into hospitals.
Concurrently, to accelerate the integration of innovative medical devices into clinical practice, the *Measures* advocate for joint initiatives between medical institutions and enterprises to establish training centers and application demonstration centers for innovative devices. The document explicitly pledges support for collaborative projects between medical institutions and enterprises aimed at promoting and applying high-end national medical equipment. Additionally, it outlines a coordinated strategy to establish two high-end medical equipment training and demonstration bases—one in the south and one in the north—to showcase achievements in the innovation and high-quality development of high-end medical equipment.
Moreover, the structural framework—or "backbone"—of the medical device industry ecosystem is currently being constructed. The *Measures* emphasize support for the development of high-quality industrial spaces dedicated to medical devices. Simultaneously, by issuing specific challenges regarding critical components and raw materials within the industry supply chain, the *Measures* encourage individual enterprises and consortia to undertake "challenge-based" R&D and collaborative innovation. Financial support—capped at 30 million RMB and calculated as no more than 30% of the approved R&D investment—will be provided to foster collaboration between upstream and downstream enterprises, thereby enhancing the resilience of the supply chain.
In alignment with digital trends, the *Measures* designate digital empowerment as a pivotal engine for the upgrading of the medical device industry. The document proposes accelerating the construction of high-quality datasets within the healthcare sector and refining policies governing data circulation in the medical and health fields. It encourages enterprises to establish data governance service platforms within designated pilot zones for fundamental data systems, and supports medical device companies in collaborating with large language model (LLM) developers to create and deploy industry-specific LLMs. Projects that attain a level of excellence recognized as "domestically first-class" or "internationally leading" will be eligible for financial support of up to 30 million RMB, based on their computational power costs.
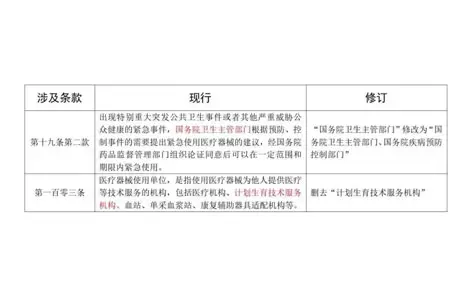
 March 02, 2026
March 02, 2026
 March 04, 2026
March 04, 2026
 March 04, 2026
March 04, 2026
 March 04, 2026
March 04, 2026Encountering problems or challenges? Please contact us!